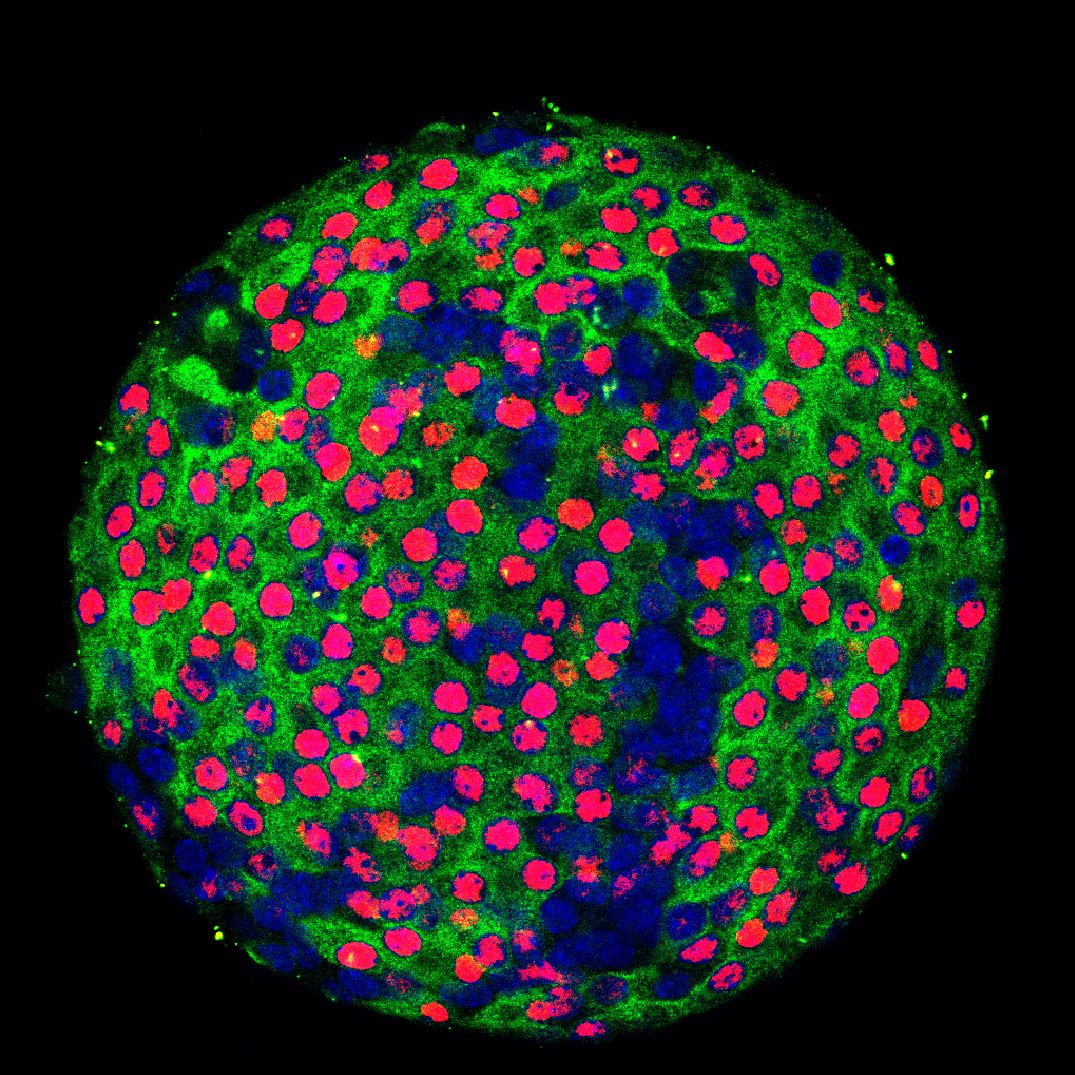
We are interested in the pancreatic beta cell – a cell whose relative or absolute deficiency results in either type 2 or type 1 diabetes mellitus. These cells are uniquely adapted to produce insulin and also to secrete it in a timely manner. In type 1 diabetes, state of the art insulin pumps and continuous glucose monitors are still poor substitutes for the beta cell in terms of blood sugar control, comfort and convenience. In type 2 diabetes, beta dysfunction is not well treated by current medical treatments (including exogenous insulin) as progressive increases in blood sugar are the rule.
To replace beta cells and/or improve beta cell function, we propose that understanding the molecular circuitry of the beta cell is crucial. After all, one cannot fix or build a machine without a blueprint. While some players are well known, we propose that there are many more genes involved in the complex process of insulin production and secretion and that these may represent novel avenues for therapeutic intervention in diabetes. Using whole genome RNA interference screens, we have identified many novel regulators of the insulin promoter and we are studying these novel components of the beta cell insulin production circuit in mouse models.
Current projects:
1) Mitochondrial dynamics in the beta cell -- It turns out that mitochondria are not a set of kidney bean shaped organelles. They fuse, divide, and move. We are studying the role of dynamics in beta cell function.
2) Regulation of the unfolded protein response by sprouty proteins -- Mammalian sprouty genes are known as developmental regulators of growth factor receptors. We believe they may be important in the beta cell and are using mouse models to understand how Spry's could regulate beta cell function.
3) Identification of targets to improve human beta cell transplantation -- Beta cell transplantation can potentiall cure type 1 diabetes, but the process is not efficient. We are finding new factors that could improve beta cell graft survival.
Funding: